COVID-19 vaccine FAQs
How do the COVID-19 vaccines work
The Pfizer and Moderna vaccines are both mRNA vaccines. This is a new type of vaccine that has not been used before. mRNA is the genetic material of the coronavirus. A small piece of the coronavirus mRNA that codes for its harmless ‘spike protein’ is injected. Our cells take-up this small piece of mRNA and use the code to manufacture the spike protein using our own cellular machinery. The protein is placed on the cell surface. Our immune system recognizes this spike protein as foreign and mounts an immune response against it. The end product of this immune response is antibodies against the spike protein. These antibodies help prevent infection from the actual coronavirus.
This is a very abbreviated and simplified description of the vaccine. If you are interested in learning more, the CDC has a nice summary of the process and a more detailed explanation below.
CDC.gov – Understanding mRNA vaccines
CDC.gov – Understanding how COVID-19 vaccines work
Does the coronavirus mRNA interact with our DNA?
No. The mRNA does not interact, affect, or get integrated in our own DNA in any way!
Is the vaccine safe?
Yes, the vaccine is safe. A group of more than 20 independent vaccinologists, immunologists, pediatricians, and other medical professionals thoroughly reviewed the data from the Pfizer and Moderna clinical trials. After that discussion, the group felt that the benefits of the vaccine far outweigh the risks of the vaccine and voted to approve both vaccines. Based on this review, the FDA granted Emergency use Authorization (EUA) for both vaccines. (note: I watched the FDA hearing personally and was very convinced that the vaccines are both safe and effective).
Here is a more in-depth discussion of the FDA EUA process:
FDA.gov – Emergency Use Authorization (EUA) explained
Are there side effects
Yes. As with most vaccines side effects are possible. The most common side effects for both vaccines are pain, swelling, and/or redness at the injection site. Fever, headache, and fatigue were also reported in a much smaller number of people. There are other very rare side effects, including a severe allergic reaction.
The CDC and the vaccine manufacturers have been very transparent with their data and the vaccine approval process. The FDA and manufacturers continue to collect data about safety and side effects. For more details, follow these links:
CDC.gov – Information on the Pfizer/BioNTech vaccine
CDC.gov – Information on the Moderna vaccine
CDC.gov – COVID-19 vaccine and severe allergic reactions
CDC.gov – What to expect after the COVID-19 vaccine
CDC.gov – Local, systemic, and allergic reactions to the Pfizer COVID-19 vaccine
Is the vaccine effective
Yes. The data shows that the both vaccines are very effective at preventing infection with the coronavirus and preventing serious illness with COVID-19. There is not enough data to determine if it helps prevent spread of the coronavirus from a vaccinated person to a susceptible person – which is why you should still wear a mask even after being vaccinated!
- How long does it take to work? – Both vaccines are two doses, separated by about three or four weeks. There is a high level of immunity starting about 1 week after the second dose.
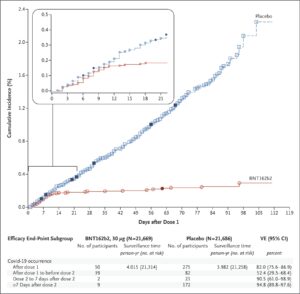
- How it was deemed to be effective? – A phase 3 clinical trials was conducted where about 50% of participants got the vaccine and 50% got placebo. COVID-19 infection rates were compared in the two groups. Those that got the vaccine were FAR less likely to contract COVID-19 (see the graph below)
- How long does immunity last? – We do not know. More studies are being conducted to answer this question.
Who will get the vaccine?
The vaccine is being distributed from the Federal Government to State Governments. Vaccine distribution decisions are made at the state and local level. This link shows Maryland’s distribution plan and what groups of people will be prioritized. Group 1A is currently receiving vaccines which includes front line health care workers and nursing homes.
MDH.gov – Vaccine deployment in Maryland
MDH.gov – Covidlink
Will the providers/staff at Potomac Pediatrics get the vaccine?
Yes! Enthusiastically, yes! In fact, just this week we received our invitation to get the COVID-19 vaccine through the Montgomery County Health Department. Many of our staff have gotten or will soon get their first dose of the vaccine!
Should kids get the vaccine? When will this happen?
Not yet. First, the Pfizer vaccine has EUA for people 16 years and older and the Moderna vaccine has EUA for 18 years and older. There is not currently enough data in the pediatric population to recommend its use in children. Second, children and teens are at far lower risk for serious COVID-19 illness so are not being prioritized for the vaccine.
Pfizer is currently enrolling for a Phase 3 clinical trial for children between 12-16 years old. It is expected that this data may be ready for analysis in late spring or early summer. We hope and expect that the vaccine will be proven similarly safe and effective in this age group. We will provide updates on this timeline as we receive them.
For those teens and young adults older than 16/18 years old (depending on the vaccine) we do recommend that they get the COVID-19 vaccine when it is their turn.
Will we have the vaccine at Potomac Pediatrics?
We don’t know (but hope so). We have filled out all of the necessary paperwork to get the COVID-19 vaccine when available and approved. We have no additional information on if/when/where/how the vaccine will be given to children at this time.
Can I get the vaccine if I am Breastfeeding
Breastfeeding mothers were not specifically studied in the vaccine trials so less data of safety and efficacy is currently available. However, The American College of Obstetricians and Gynecologists (ACOG) and other organizations have recommended that lactating women should have equal access to the vaccine and that breastfeeding is not contraindicated after getting the vaccine.
ACOG.org – Clinical Guidance for COVID-19 vaccine
CDC.gov – COVID-19 vaccine considerations for pregnant and breastfeeding women
Can I get the vaccine if I am pregnant?
Similar to breastfeeding, the vaccine studies did not look specifically at safety or efficacy of the vaccine in pregnant women. However, ACOG has recommended that “COVID-19 vaccines should not be withheld from pregnant individuals who meet criteria for vaccination based on ACIP-recommended priority groups.” If you have specific questions, we recommend that you speak with your OB/GYN.
Should I get the vaccine if I have already had COVID-19?
Yes. It is unclear how long natural immunity after having COVID-19 lasts. So, those that have been previously infected should still get the vaccine when it is their turn.
Bottom line, should I get the vaccine when it is my turn?
YES! Unless you have a specific contraindication to the vaccines, all of us at Potomac Pediatrics believe in the power of science, data, and vaccines. We think it is important for your family’s personal health and public health that you get the vaccine when it is your turn!
CDC.gov – Benefits of COVID-19 vaccine
CDC.gov – COVID-19 vaccine FAQs











Leave a reply